Urs Giger, PD, Dr. med. vet., MS, FVH, DACVIM, DECVIM
Hereditary disorders may affect any blood cells or plasma proteins. For many hereditary blood diseases, the biochemical basis has been elucidated; for some, the specific molecular genetic defect has recently been identified. In fact, erythrocytic pyruvate kinase deficiency was the first biochemical defect characterized in companion animals, and hemophilia B was the first molecular defect determined in domestic animals. On the other hand, the precise cause of cyclic hematopiesis in Gray Collies still needs to be discovered despite extensive research over the past 30 years. Overall the study of these hereditary blood disorders has greatly contributed to the better understanding of blood cells and protein functions.
Hereditary blood disorders may be classified into erythrocytic defects, bleeding disorders, and immunodeficiencies, although some overlap exists. Several hereditary disorders not only affect a specific function of the hematopoietic system, but also involve other organs such as bone, muscle, and hair coat, thereby forming characteristic clinical syndromes. Finally, an emerging group of genetic predispositions to infections, immune-mediated, neoplastic diseases are being recognized. Some primary immunodeficiencies (Leukocyte adhesion deficiency in Irish setters) are well defined, whereas for others only a breed predilection to a particular infection suggests a genetic basis, for instance avian tuberculosis in Bassets and leishmaniasis in Foxhounds. Similarly, breed predispositions have been recognized for certain hematologic immune-mediated diseases (immune-mediated hemolytic anemia in Cocker Spaniels) and hematopoietic cancers (lymphoma in Golden retrievers and malignant histiocytosis in Bernese Mountain dogs) without knowing the molecular basis.
With few exceptions, hereditary blood disorders are autosomal recessively inherited; hemophilia A and B in many breeds of dogs and cats and severe combined immunodeficiency in Bassets and Corgis represent the only X-chromosomal recessive traits. Each defect occurs relatively rarely, although certain breeding practices, popular sire, and founder effects may result in widespread and common occurrences. The true mutant gene frequency has rarely been determined to limit the further spread of these hereditary disorders, it is pivotal to not only recognize affected animals, but also carriers that can pass on the mutant gene. Accurate biochemical and molecular genetic screening tests have been developed for many diseases, but their application in companion animals has lagged behind.
Therapeutic options for hereditary blood diseases are limited. Animals with primary immunodeficiencies may benefit from antibiotics, albeit their response may only be partial or transient. Plasma transfusions may supplement bleeding animals with coagulopathies. Experimental allogeneic bone marrow transplantation can correct defective blood cells and gene therapy has been attempted. However, it is much more important to control further spread of the disease causing gene by screening breeding animals when tests are available. Although acquired causes, such as infections, immune disorders, intoxication, blood loss, and chronic organ failure, are the main causes for anemia, hereditary blood disorders leading to anemia are also important in clinical practice. They are particularly germane differential diagnoses in animals with Coombs' negative hemolytic anemias without apparent cause. Several hereditary erythrocytic defects have been reported in companion animals, and much new information has emerged over the past decade. In fact, some anemias have been so extensively characterized that the clinical signs to the molecular basis of the erythrocyte defect are known, thereby offering an opportunity to make a precise diagnosis in clinical practice and to prevent these disorders in future generations.
Hereditary anemias
Inherited erythrocyte defects form a large heterogeneous group of diseases. Each erythrocyte disorder is observed only rarely, although a particular defect may occur frequently within a family or breed. If the same disorder is recognized in several breeds, it is likely caused by different mutations of the same gene. The mode of inheritance is autosomal recessive, with the exception of feline porphyria which is inherited by a dominant trait. These disorders have been classified into four groups: 1) heme defects and hemoglobinopathies, 2) membrane abnormalities, 3) cytosolic enzyme deficiencies, and 4) production and maturation defects.
In contrast to the common occurrence of such hemoglobin disorders as thalassemia and sickle cell anemia in humans, no hemoglobinopathies have been documented in dogs and cats. Isolated cases of methemoglobinemia associated with methemoglobin/cytochrome b5 reductase deficiency were found among dogs of various breeds and domestic shorthair cats, but this deficiency results in polycythemia rather than anemia. Defects of heme synthesis known as porphyrias have been reported in anemic Siamese and domestic shorthair cats with pigmented and pink-fluorescent teeth and bones.
Elliptocytosis and microcytosis resulting from a deficiency of the protein band 4.1, which strengthens the interaction between spectrin and actin in the cytoskeleton, has been characterized at the molecular level in an inbred, nonanemic mongrel dog. Other presumed membrane abnormalities include: stomatocytosis in Alaskan malamutes, a Dutch breed with gastritis, and miniature Schnauzers; nonspherocytic anemia in Beagles; and increased osmotic fragility of erythrocytes in an English Springer spaniel and Abyssinian and Somali cats.
Deficiencies of the two key regulatory glycolytic enzymes result in distinctly different forms of hemolytic anemia. The classic pyruvate kinase (PK) deficiency initially reported in Basenjis is now seen in several other canine breeds and in cats. Phosphofructokinase (PFK) deficiency is frequently reported in English Springer spaniels and has also been observed in a Cocker spaniel and a mixed-breed dog.
Erythrocytic Disorders (Examples)
|
Erythrocyte membrane defects (hemolysis) |
|
Elliptocytosis |
Mixed breed dog |
|
|
Stomatocytosis |
Malamute (chondrodysplasia)
Miniature Schnauzers |
|
|
Spherocytosis |
Retriever |
|
|
? Osmotic fragility |
Abyssinian and Somali cats |
|
|
Eythroenzymopathies (hemolysis) |
|
Phosphofructokinase deficiency |
English Springer Spaniel
Cocker Spaniel, mixed breed |
|
Pyruvate kinase deficiency
(dogs w/ osteosclerosis) |
Basenji, Beagle, Eskimo Toy
West Highland White Terrier
Cairn Terrier, Dachshund
Abyssinian and Somali cats |
|
Methemoglobin reductase deficiency |
Many canine breeds, cats |
|
No hemoglobinopathies |
|
|
Erythrocyte production defects (non-regenerative) |
|
Cobalamin malabsorption
(non-regenerative anemia) |
Giant Schnauzer, Beagle,
Border Collie & Austr. Shepherd |
|
|
Macrocytosis |
Miniature Poodle |
|
|
Microcytosis |
Japanese breeds (no anemia) |
|
Hereditary Bleeding Disorders
|
Disorder |
Breed |
BMBT |
ACT PTT |
PT PIVKA |
Remarks |
|
Coagulopathies |
|
|
|
|
|
|
Prothrombin deficiency |
Cocker Spaniel, Boxer |
N |
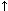
|
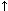
|
|
|
Factor VII deficiency |
Beagle, Malamute |
N |
N |
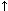
|
Colony of beagles |
|
Factor VIII deficiency (Hemophilia A) |
Many breeds, cats (x-linked) |
N |
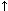
|
N |
Mild to severe forms |
|
Factor IX deficiency (Hemophilia B) |
Many breeds, cats (x-linked) |
N |
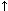
|
N |
DNA tests for some available |
|
Factor X deficiency |
Cocker Spaniel |
N |
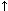
|
N |
|
|
Factor XI deficiency |
Kerry Blue terriers, Great Pyrenese, English Springer |
N |
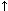
|
N |
Late posttraumatic |
|
Factor XII deficiency |
Cats |
N |
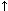
|
N |
No bleeding |
|
Vitamin K dependent coagulopathy |
Devon Rex |
N |
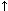
|
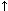
|
|
|
Von Willebrand Disease |
|
|
|
|
|
vWD Type 1 (common) Complete vWF deficiency; <1% plasma vWF levels |
Doberman, many others |
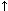
|
N |
N |
DNA test for Doberman, Manchester & Cairn terrier |
|
vWD Type 2 (rare) Disproportionally low vWF activity; deficiency of high molecular weight multimers |
German shorthair pointer |
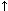
|
N |
N |
DNA test |
|
vWD Type 3 (severe) Complete vWF deficiency; <1% plasma vWF levels |
Scottish terriers, Shelties, Chesapeake Bay retriever |
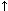
|
N |
N D |
NA test for Sheltie, Kooiker, Scottish terrier |
|
|
|
|
|
|
|
|
Platelet Dysfunctions |
|
|
|
|
|
Delta storage pool disease |
Cocker spaniel |
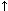
|
N |
N |
High ATP/ADP ratio |
|
Glanzmann thrombasthenia |
Otterhound, Great Pyrenese |
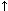
|
N |
N |
GP IIb/IIIa deficiency |
|
Chediak Higashi |
Smoke Persians |
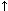
|
N |
N |
WBC granulation |
|
Thrombopathies (others) |
Basset hound, Spitz, Boxer, Labrador, etc., DSH cats |
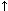 / N / N
|
N |
N |
Signaling defect or unknown |
N-normal, 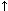 -prolonged
-prolonged
Primary or Hereditary Immunodeficiencies of Dogs and Cats
|
Disease |
Inherit |
Breeds |
Defect |
Characterization |
|
DOG |
|
|
|
|
|
Ciliary dyskinesia(Immotile cilia syndrome) |
AR |
Many breeds |
Functional/morphologic cilia abnormalities |
Rhinosinusitis, broncho pneumonia, bronchi ectasis, +/-situs inversus |
|
Bactericidal neutrophil defect |
U |
Doberman |
Unknown |
Upper respiratory infections, ciliary dyskinesia not excluded |
|
Cyclic hematopoiesis (Cyclic neutropenia) |
AR |
Gray Collie |
Hematopoietic growth factors |
Severe neutropenia every 12-14 days, reactive amyloidosis |
|
Leukocyte adhesion Deficiency (CLAD) |
AR |
Irish Setter, Red & White Setters |
CD11/18 deficiency ß chain (CD18) deficiency |
Severe leukocytosis, limited pus formation, lack of neutrophil adhesion |
|
Complement component 3 C3 deficiency) |
AR |
Brittany Spaniel |
C3 deficiency |
Pyogenic infections, phagocytosis |
|
Selective cobalamin malabsorption |
AR |
G. Schnauzer, Border collie, Beagle, Aust. Shepherds |
Ileal cobalamin receptor defect |
Weight loss, inappetence, leukopenia with hyper- segmentation, methylmalonic aciduria |
|
? Susceptibility to avian mycobacteriosis |
U |
Basset hound |
RAMP deficiency? |
Systemic avian tuberculosis, toxoplasmosis & neosporosis |
|
? Susceptibility to pneumocystitis pneumonia |
AR |
Dachshund |
Unknown |
Pneumocystitis pneumonia |
|
Pelger-Huet anomaly |
AD |
Australian Shepherd, foxhound |
Unknown |
No immunodeficiency, hyposeg. granulocytes |
|
? Susceptibility to fungal & rickettsial infect.; pyoderma |
U |
German shepherd |
Macrophage?, T- cell? |
Severe ehrlichiosis, RMSF disseminated aspergillosis, deep pyoderma |
|
X-SCID - X-linked severe combined immunodeficiency |
XR |
Basset hound, Card. Welsh Corgi |
Common gamma chain of IL-2 and other cytokines |
Severe bacterial/viral infections, no IgG & IgA |
|
Severe combined immunodeficiency |
AR |
Jack Russell terrier |
DNA-protein kinase catalytic subunit |
Severe serum immunoglobulin deficiency, hypoplasia of lymphoid tissues |
|
Selective IgA deficiency |
U |
Beagle, Shar Pei, German S |
IgA deficiency |
Respiratory & gastrointestinal infections |
|
Thymic abnormal. & dwarfism |
U |
Weimaraner |
Unknown |
Reduced growth, thymosin responsive |
|
Recurrent infections |
U |
Weimaraner |
Reduced IgG |
Pyoderma, abscessation, bleeding tendency |
|
Combined immunodeficiency |
U |
Shar Pei |
T-cell, B-cell, low IL-6 & IL-2 |
Skin, respiratory & GI infections |
|
|
Amyloidosis |
U |
Shar Pei |
Elevated IL-6 |
Arthritis, amyloidosis, renal failure, hepatic rupture, hypoproteinemia |
|
|
Lethal acrodermatitis |
AR |
Bull Terrier |
Zinc metabolism defect |
Zinc deficiency, hyperkeratosis |
|
|
? Susceptibility to parvoviral infection |
U |
Rottweiler Doberman |
Unknown |
Parvovirus infection |
|
|
Vaccine-induced immune disturbance |
U |
Akitas |
Unknown |
Variable meningitis, polyarthritis, amyloidosis |
|
|
CAT |
|
|
|
|
|
|
Hypotrichosis congenital and thymic atrophy |
AR |
Birman |
Unknown |
Nude kittens, neonatal death, no thymus |
|
|
Leukocyte granulation |
U |
Birman |
Unknown |
No immunodeficiency, acidophilic granules |
|
|
Pelger-Huet anomaly |
AD |
Domestic shorthair |
Unknown |
No immunodeficiency, hyposegmentation |
|
|
Chédiak-Higashi syndrome |
AR |
Persian |
Unknown |
No immunodeficiency, granules in phagocytes, bleeding tendency |
|
|
Reactive (AA) amyloidosis |
U |
Abyssinian |
Unknown |
Reactive (AA) amyloidosis, renal failure |
|
|
|
|
|
|
|
|
|
AR = autosomal recessive; U = unknown; XR = X-linked recessive; AD = autosonal dominant.